We Bridge the Gap Between "Clinical Data" and "Commercial Understanding."
We do not "dumb down" science; we translate it. Our teams understand the difference between Efficacy and Effectiveness, and we design to specific MLR (Medical, Legal, Regulatory) standards.

Mechanism of Action (MOA) Visualization
We translate complex Pathophysiology and Mechanism of Action into executive-ready narratives. We visualize how your molecule interacts with the target at a cellular level, making the 'Unmet Need' and 'Value Proposition' obvious to investors, HCPs, and Payers.

Ad-Comms & Regulatory Defense
For FDA Advisory Committees and critical regulatory panels, clarity is a defense strategy. We build "Audit-Ready" presentations that allow your team to navigate Q&A sessions with precision, ensuring the data supports the argument instantly.

Scalable Production Infrastructure: Handling 300+ Slides/Month
For global strategy firms and brand teams, we provide a dedicated 'Shadow Shift'—a secure, high-volume production engine capable of managing 10-12 workstreams simultaneously with 24-hour delivery.
With increasing scientific data complexity, fragmented stakeholder ecosystems, and regulatory sensitivity, pharma and life sciences-focused communication is challenged. We help interpret the latent narratives to support more informed healthcare decision
Trusted by



Your data is your IP. We treat it with the security it demands
MLR Fluency
Our clinical data visualization workflow includes built-in referencing and annotation layers. We ensure every pharma presentation is engineered for speed-to-market without compromising scientific accuracy.
Global Experience
We have supported major players like Abbott and Glenmark, delivering high-stakes presentations that respect scientific accuracy.
Data Security
Our workflows are encrypted and NDA-protected, ensuring your clinical findings remain confidential until publication.
Clinical Integrity & Specialized Visual Engineering.
We engineer specialized clinical visuals that preserve scientific rigor while making leadership reviews faster, clearer, and easier to act on.
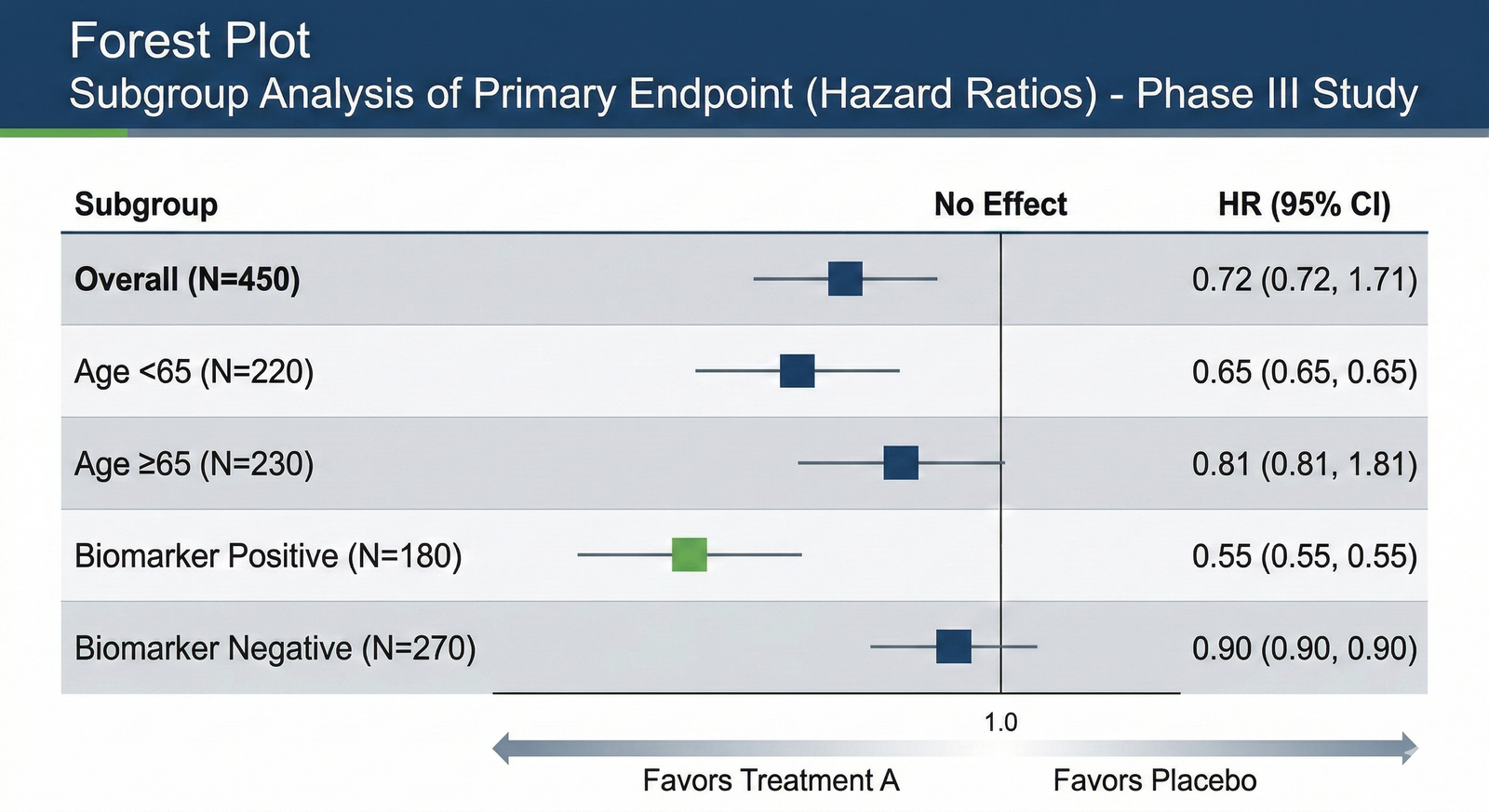
Precision Forest Plots (Hazard Ratios)
We do not just align boxes; we accurately plot subgroup analyses (Hazard Ratios).
For high-stakes clinical readouts, we ensure subgroup analyses, including Age, Gender, and Biomarker Status, are plotted with 100% Data Integrity.
We understand the significance of Hazard Ratio (HR) placement and 95% Confidence Intervals.
We move beyond automated tool outputs to create custom, boardroom-ready visuals that clearly communicate efficacy across all pre-specified subgroups, accelerating executive decision-making.
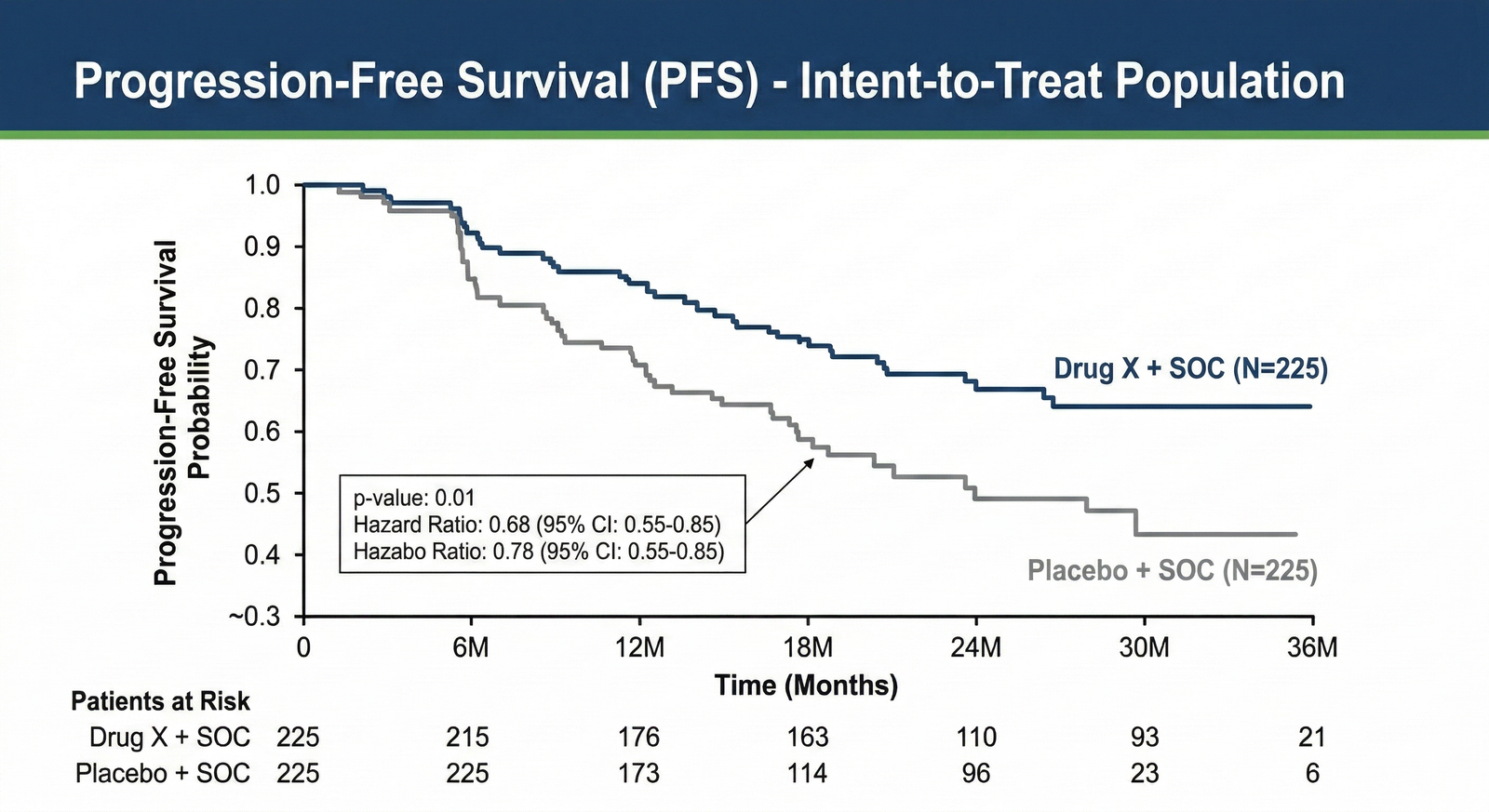
Kaplan-Meier Survival Analysis
We understand 'Patients at Risk' and 'p-value' hierarchy.
We transform time-to-event data (PFS/OS) into intuitive, rigorous visual narratives.
Validated p-value & HR Annotations: Ensuring key statistical outputs are instantly legible for leadership reviews.
Patients at Risk Tables: Critically, we always include this essential data component at the bottom, eliminating the need for stakeholders to cross-reference multiple documents.
Censoring Indications: Precise marking to maintain scientific transparency while improving overall visual hierarchy.
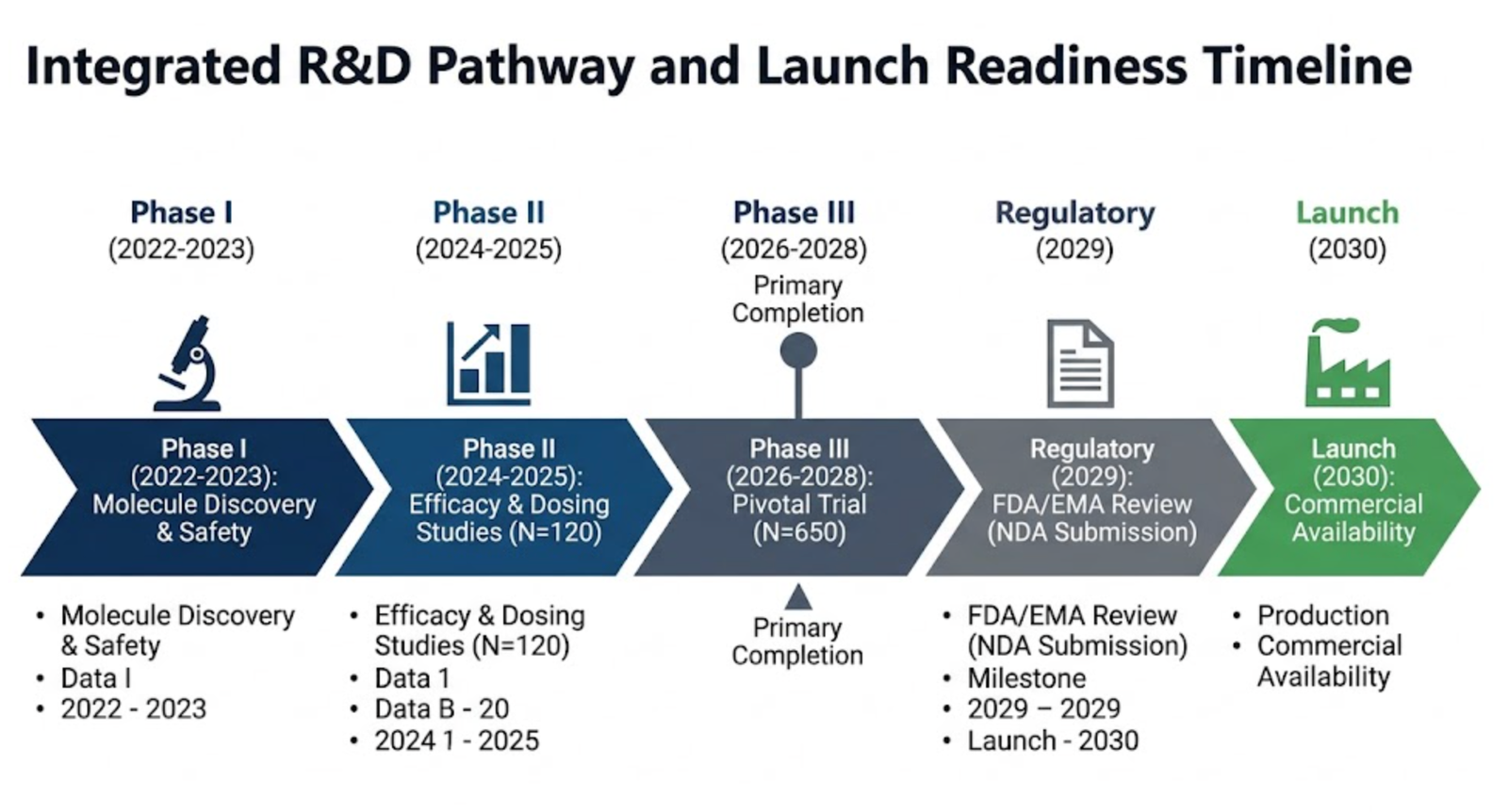
Integrated Clinical Trial & Launch Readiness Timelines
We support Lifecycle Management, not just the readout.
For global strategy and commercial teams, we provide a unified view of your entire R&D pipeline.
We architect complex multi-year timelines, visualizing enrollment targets, regulatory milestones (NDA/BLA submissions), and primary completion dates.
This asymmetric operational advantage accelerates 'Launch Velocity' by ensuring all stakeholder teams, from Medical Affairs to Field Force, are aligned on the critical path to commercialization.
